Introduction
Guillain-Barré syndrome (GBS) is a condition characterised by the autoimmune destruction of the peripheral sensory system[1]. It can involve sensory, motor, and autonomic nerves. There occurs temporary inflammation and demyelination of peripheral nerve myelin sheaths resulting in axonal degeneration. It is the most common cause of quickly progressive flaccid paralysis. It is believed to be one of a number of related conditions, all sharing a similar underlying autoimmune abnormality, as a group known as anti-GQ1b IgG antibody syndrome.
Chronic inflammatory demyelinating polyneuropathy (CIDP) is considered the chronic counterpart to Guillain-Barré syndrome.[2]
[3]
Aetiology
Most cases of GBS are preceded by upper respiratory tract infections or diarrhoea one to three weeks prior to their onset. The most common infections causing GBS include Campylobacter jejuni, Mycoplasma pneumoniae, and Cytomegalovirus[4]. Campylobacter jejuni is responsible for about a third of GBS cases, and the ensuing GBS is usually more severe than that due to other causes[4]. Recent infection with Campylobacter jejuni is strongly associated with developing AIDP or AMAN[5]. Molecular imitation by the bacterial agents is thought to cause the autoimmunity with the development of anti-GQ1b IgG antibodies. GBS often presents (70% of patients) within 1-6 weeks of prior illness[6].
Alternative predisposing factors include recent surgery, lymphoma, and systemic lupus erythematosus (SLE).[2]
Epidemiology
- Incidence: The annual incidence of GBS in the USA is 1.2-3 per 100,000 inhabitants[7]; GBS has been reported throughout the world. Most studies show annual incidence figures similar to those in the United States[8].
- Age: The annual mean rate of hospitalizations in the United States related to GBS increases with age, being 1.5 cases per 100,000 population in children under 15 years of age, and peaking at 8.6 cases per 100,000 population in 70-79 year olds[9]
- Men are more likely to develop Guillain–Barré syndrome than women; the relative risk for men is 1.78 compared to women[10].
Introducing PAI - Your AI Clinical Partner in Physiotherapy
Enhance your clinical reasoning, minimise paperwork and maximise patient interaction
Pathophysiology
The pathophysiology of GBS is complex. GBS is considered to be an autoimmune disease triggered by a preceding bacterial or viral infection. Campylobacter jejuni, cytomegalovirus, Epstein-Barr virus and Mycoplasma pneumoniae are commonly identified antecedent pathogens.
The body's immune system begins to attack the body itself,[11] The immune response causes a cross-reaction with the neural tissue. When myelin is destroyed, destruction is accompanied by inflammation. These acute inflammatory lesions are present within several days of the onset of symptoms. Nerve conduction is slowed and may be blocked completely. Even though the Schwann cells that produce myelin in the peripheral nervous system are destroyed, the axons are left intact in all but the most severe cases. After 2-3 weeks of demyelination, the Schwann cells begin to proliferate, inflammation subsides, and re-myelination begins.
Clinical Presentation
Typically, GBS symptoms present 2-4 weeks after a relatively benign gastrointestinal or respiratory illness. The first neurological sign of GBS is usually paresthesias of the toes, followed by a sudden onset (hours to days) of symmetrical, progressive bilateral weakness and sensory loss distal to proximal throughout the body. This dysfunction may impact the muscles of respiration, and even cranial nerves cranial nerves. In most forms of GBS, there is a greater loss of motor function than sensory function. [12]
Pain is an often under-recognised symptom of GBS[13]. In a study following 55 patients with GBS, 89% reported pain, with 47% grading it as severe[14]. Another study following 223 patients with GBS found that 55% experienced pain, with 70% experiencing pain before the onset of weakness[15]. Pain was described as deep and achy in the lower back and legs, and patients also described dysesthesia in their extremities[14]. The pain improved with time, but the dysesthesia remained in a small group of patients[14]. Another study suggested that pain may last up to 2 years for some patients[16].
Two types of pain can be classified in GBS. The first type of pain is more acute, beginning before onset of muscle weakness and lasts until discharge from hospital. It presents as mainly radicular, muscle pain and dysesthesia of the extremities. The second type is observed during the chronic stage - patients complain of dysesthesia, muscle pain and arthralgia in the limbs; and these are associated with weakness and disability[13].
GBS can be split up into two types according to electrophysiologic and pathologic features: the demyelinating type, and the axonal type[17]. Subtypes in line with these include:
- Acute inflammatory demyelinating polyradiculoneuropathy (AIDP): most common form (60-90%)
- Axonal subtypes: acute motor axonal neuropathy (AMAN) (historically Chinese paralytic syndrome); acute motor-sensory axonal neuropathy (AMSAN)
- Regional GBS syndromes: Miller Fisher variant (MFS/MFV), characterised by ataxia, ophthalmoplegia, and areflexia without weakness, anti-GQ1b antibodies are present in most cases; polyneuritis cranialis.[7]
Symptoms Progression
Antecedent Illness: Upper respiratory and gastrointestinal illnesses are the most commonly reported conditions[18]. Symptoms generally have resolved by the time the patient presents with the neurological condition.
GBS Symptom progression: The mean time to the peak of symptoms is 12 days (from 1st neurological symptoms), with 98% of patients reaching a peak by 4 weeks. A plateau phase of persistent, unchanging symptoms then ensues, followed days later by gradual symptom improvement[19]. Recovery usually begins 2-4 weeks after the progression ceases[20]. The mean time to clinical recovery is 200 days.
Overall, most patients with GBS do well, with 85% of patients recovering independent ambulation; however, 20% of patients continue with morbidity[1]. More than 80% of patients achieve independent ambulation within six months[21].
Differential Diagnosis
- Acute peripheral neuropathies
- Toxic: thallium, arsenic, lead, n-hexane, organophosphate
- Drugs: amiodarone, perhexiline, gold
- Alcohol
- Porphyria
- Systemic vasculitis
- Poliomyelitis
- Diphtheria
- Tick paralysis
- Critical illness polyneuropathy
- Disorders of Neuromuscular Transmission
Diagnostic Procedures
Guillain-Barre syndrome (GBS) is considered a clinical diagnosis and a diagnosis can be made at the bedside in most cases. For atypical cases or unusual subtypes, ancillary testing can be useful.
These include:
- Cerebrospinal fluid investigation: It will elevated at some stage of the illness but remains normal during the first 10 days. There may be lymphocytosis (> 50000000 cells/L).
- Electrophysiological studies: it includes nerve conduction studies and electromyography. They are normal in the early stages but show typical changes after a week or so with conduction block and multifocal motor slowing, sometimes most evident proximally as delayed F-waves.
The only way to classify a patient as having the axonal or nonaxonal type is electrodiagnostically. - Further investigative procedures can be undertaken to identify an underlying cause
For example:- Chest X-ray , stool culture and appropriate immunological tests to rule out the presence of cytomegalovirus or mycoplasma
- Antibodies to the ganglioside GQ1b for Miller Fisher Variant.
- MRI
- Lumbar Puncture: Most, but not all, patients with GBS have an elevated CSF protein level (>400 mg/L), with normal CSF cell counts. Elevated or rising protein levels on serial lumbar punctures and 10 or fewer mononuclear cells/mm3 strongly support the diagnosis.
Prognosis
Guillain-Barré syndrome can be a devastating disorder because of its sudden and unexpected onset. In addition, recovery is not necessarily quick. Typically, improvement occurs after a number of weeks to months, although there is significant mortality (3-10%).[2]
The majority of people recover completely or nearly completely[22]. However, some have mild residual effects such as foot drops or abnormal feeling in the feet and hands that persists for two years or more. Persistent fatigue and pain may present. Fewer than 15 percent have a substantial long-term disability severe enough to need a cane, walker or wheelchair.
Predictive factors for poor outcome for GBS include[23][24][25]:
- Older age
- Cranial nerve impairment
- Recent surgery
- Elevated level of liver enzymes
- Diabetes
- High blood pressure at admission
- Uroschesis
- Fever
- Ventilator support during hospitalization
- Disorder of consciousness
- Absence of preceding respiratory infection
Management
There is no known cure for Guillain-Barré syndrome, but therapies that lessen the severity of the illness and accelerate the recovery in most patients exist. GBS is primarily managed with Intravenous (IV) immunoglobulin or plasmapheresis along with supportive measures, which can hasten recovery.[2]
Further Medical Management Can Be Done According to the Symptoms and the Complications
- Supportive Care
- ICU monitoring
- Basic medical management often determines mortality and morbidity.
- Ventilatory Support
- Atelectasis leads to hypoxia.
- Hypercarbia later finding; arterial blood gases may be misleading.
- Vital capacity, tidal volume and negative inspiratory force are the best indicators of diaphragmatic function.
- Progressive decline of these functions indicates an impending need or ventilatory assistance. Mechanical ventilation usually required if VC drops below about 14 ml/kg; ultimate risk depends on age, the presence of accompanying lung disease, aspiration risk, and assessment of respiratory muscle fatigue.
- Atelectasis treated initially by incentive spirometry, frequent suctioning, and chest physiotherapy to mobilize secretions.
- Intubation may be necessary for patients with substantial oro-pharyngeal dysfunction to prevent aspiration.
- Tracheostomy may be needed in patients intubated for 2 weeks who do not show improvement.
- Autonomic Dysfunction
- Autonomic dysfunction may be self-limited; do not over-treat.
- Sustained hypertension managed by angiotensin-converting enzyme inhibitor or beta-blocking agent. Use short-acting intravenous medication for labile hypertension requiring immediate therapy.
- Postural hypotension treated with fluid bolus or positioning.
- Urinary difficulties may require intermittent catheterization.
- Nosocomial Infections Usually Involve Pulmonary and Urinary Tracts.
- Occasionally central venous catheters become infected.
- Antibiotic therapy should be reserved for those patients showing clinical infection rather than the colonization of fluid or sputum specimens.
- Venous Thrombosis Due to Immobilization Poses a Great Risk of Thromboembolism
- Prophylactic use of subcutaneous heparin and compression stockings.
Physiotherapy Management
Studies have shown that exercise can positively influence outcomes such as mobility, fatigue levels and even mental function in GBS patients[26][27]. One study suggests that high-intensity exercise induces greater improvement in FIM motor scores, compared to lower intensity[28]. However, over-exercise for patients with GBS of partially denervated muscles can cause further damage, including a loss of functioning motor units[29], hence the need to closely monitor exercise intensity to avoid over-exercising partially denervated motor units[30][31].
Aims of physiotherapy management are:
- Regain the patient's independence with everyday tasks.
- Retrain normal movement patterns.
- Improve patient's posture.
- Improve the balance and coordination
- Maintain clear airways
- Prevent lung infection
- Support joint in functional position to minimize damage or deformity
- Prevention of pressure sores
- Maintain peripheral circulation
- Provide psychological support for the patient and relatives.
A tri-phasic approach was suggested for physical therapy management when comparing to disease progression:
- Acute/Ascending Phase (first 2-3 weeks): prevention of complications of immobilization (contracture, pressure injury, etc.), supporting pulmonary function, and pain management.
- Plateau Phase: increasing upright posturing, improving pulmonary function while avoiding fatigue/overexertion, gentle stretching and active-assisted and active range of motion as tolerated for gradual improvements in mobility.
- Recovery/Descending Phase (~2-4 weeks after the plateau phase): increased upright posturing and weight-bearing, encouraging a high-intensity rehabilitation approach utilizing active resistance training and neuromuscular facilitation techniques. [32]
Respiratory Care
The common respiratory complications in the rehabilitation setting include incomplete respiratory recovery including chronic obstructive pulmonary disease, restrictive respiratory disease (pulmonary scarring, pneumonia), and tracheitis from chronic intubation and respiratory muscle insufficiency. Sleep hypercapnia and hypoxia, which worsens during sleep can be the result of a restrictive pulmonary function.[33][34]
Treatment methods are:
- Nighttime saturation records with a pulse oximeter and bilevel positive airway pressure (BiPAP) may be indicated for the patients.
- Physical therapy measures (chest percussion, breathing exercises, resistive inspiratory training) may be required to clear respiratory secretions to reduce the work of breathing.
- Special weaning protocol to prevent over-fatigue of respiratory muscles can be recommended for more severe patients with tracheostomy. Patients with cranial nerve involvement need extra monitoring as they are more prone to respiratory dysfunction.
- Patients should be encouraged to cease smoking.
- Posturally drain areas of lung tissues, 2-hourly turning into supine or side-lying positions.
- 2-4 litre anesthetic bag can be used to enhance chest expansion. Therefore, 2 people are necessary for this technique, one to squeeze the bag and another to apply chest manipulation.
- Rib springing to stimulate cough.
- After the removal of a ventilator and adequate expansion, effective coughing must be taught to the patient.
Maintain Normal Range of Movement
Gentle passive movements through full ROM at least three times a day especially at the hip, shoulder, wrist, ankle, and feet.
Use of light splints (eg. using PLASTAZOTE) may be required for the following purpose listed below:
- Support the peripheral joints in a comfortable and functional position during flaccid paralysis.
- To prevent abnormal movements.
- To stabilize patients using sandbags, and pillows.
Prevention of Pressure Sores
Change in patient's position from supine to side-lying after every 2 hours. If the sores have developed then UVR or ice cube massage to enhance healing.
Maintenance of Circulation
- Passive movements
- Effleurage massage to lower limbs.
Relief of Pain
- Transcutaneous electrical nerve stimulation
- Massage with passive ROM
- The patient can demonstrate increased sensitivity to light touch, a cradle can be used to keep the bedsheet away from the skin. Low-pressure wrapping or snug-fitting garments can provide a way to avoid light touch.
- Reassurance and explanation of what to expect can help in the alleviation of anxiety that could compound the pain.
Strength and Endurance training
Strengthening exercises can involve isometric, isotonic or isokinetic exercises, while endurance training involves progressively increasing the intensity and duration of functional activities such as walking or stair-climbing[35].
Functional training
Retraining of dressing, washing, bed mobility, transfers, and ambulation activities comprise a big part of the rehabilitation process. Balance and proprioception retraining in all these functional activities should also be included, while motor control can be achieved by doing Proprioceptive Neuromuscular Facilitation (PNF) techniques[35].
Research shows that high-intensity relative to lower intensity exercise significantly reduced disability in patients with GBS, as measured with the FIM (p<0.005, r=0.71). Overall, various types of exercise programmes improve physical outcomes such as functional mobility, cardiopulmonary function, isokinetic muscle strength, and work rate and reduce fatigue in patients with GBS[36][37].
Assistive devices
Assistive devices such as wheelchairs, walking sticks and quadrupeds should be made available to individuals if required in order to facilitate safe and effective ambulation[35].
According to Bensman (1970), the following four guidelines are to be followed for the prescription of exercises:
- Use short periods of non-fatiguing exercises matched to the patients strength.
- Progression of the exercise should be done only if the patient improves or if there is no deterioration in status after a week.
- Return the patient to bed rest if a decrease in muscle strength or function occurs.
- The objective should be directed not only at improving function but also at improving strength.
*A study of 35 patients (27 with classic GBS and 8 with acute motor axonal neuropathy [AMAN]), reported GBS-related deficits included: neuropathic pain requiring medication therapy (28 patients) *foot drop necessitating ankle-foot orthosis (AFO) use (21 patients) *locomotion difficulties requiring assistive devices (30 patients) *At 1-year follow-up, the authors found continued foot drop in 12 of the AFO patients. However, significant overall functional recovery had occurred within the general cohort[38] (LoE 1B).
Nehal and Manisha (2015) suggest a functional goal-oriented multidisciplinary rehabilitation programme for daily 1 hour sessions for 12 weeks.
Additional Read
Case Study: Guillain-Barre Syndrome (Sub-Acute)
Guillain-Barré Case Study: Marie
Guillain-Barré Case Study: David
Related articles
Acute Motor Axonal Neuropathy (AMAN), a Variant of Guillain-Barre Syndrome: A Case Study - PhysiopediaAbstract This case study documents a fictional description of a patient presenting with acute motor axonal neuropathy (AMAN), which is a subtype of Guillain-Barre Syndrome. The patient’s experience in acute care is discussed briefly, while the patients’ sub-acute and long-term rehabilitation is covered in more detail. The purpose of this case study is to highlight approaches to assessment, outcome planning, and treatment interventions in the context of a community-based rehabilitation setting during the sub-acute phase of the disease. The case study is unique as it employs an interdisciplinary approach and international classification of function, disability, and health (ICF) framework to address the pathology of the disease in a patient-specific manner. Introduction[edit | edit source] Acute motor axonal neuropathy (AMAN) is a variant type of Guillain-Barre Syndrome (GBS). AMAN is a non-inflammatory disease whereby axons of motor nerve cells are selectively targeted and destroyed by the body’s own immune system. The myelin sheath surrounding the axon is unaffected. Studies suggest that the body’s immune system specifically attacks the membrane surrounding the axon called the axolemma. AMAN is characterized by acute progressive motor weakness, areflexia, ataxia, oculomotor dysfunction and absence of sensory symptoms[1]. AMAN is typically preceded by an infection from bacteria called Campylobacter jejuni[2] or Haemophilus influenzae[3]. Prevalence of AMAN is estimated at 5% of cases in North America and Europe[1], while 30-65% of cases are located in Asia, Central and South America[4]. The prognosis involves two categories of patients undergoing rapid recovery or slower recovery after reaching the highest levels of unchanging symptoms and dysfunction, or plateau phase[5]. It is proposed that recovery is achieved through mechanisms such as terminal motor nerve axonal regeneration and collateral reinnervation processes[6]. Documented cases in literature that are relevant find absent or minor sensory deficits are present among patients with AMAN, and majority of patients have complete symptom resolution with 5-day intravenous immunoglobulin (IV Ig) treatment[7]. Patients with AMAN continue to see improvements in ambulation and functional ability up to four years post-diagnosis[3]. This brings us to the purpose of the following case study of a patient with AMAN. Physiotherapy assessment and treatment can help prevent the decline of functional status and maintain functional independence, muscle strength, posture, balance, and cardio-respiratory fitness. Moreover, it enables an individual to continue performing self-care routines and activities of daily living (ADLs). In accordance to the International Classification of Functioning, Disability and Health (ICF) framework[8], the primary objective of this case study is to elaborate on how physical therapy rehabilitation assessment and treatment interventions can aim to address body structure and function impairments, activity limitations, and participation restrictions in patients with AMAN. The case study further aims to focus on patient-centred care and an interdisciplinary approach to assessment and management of AMAN in a community-based rehabilitation setting. [9] Case Background[edit | edit source] Trish Jones, a 62 year old female, initially presented with nausea and diarrhea one week after returning home from vacation in Mexico. Due to COVID-19 travel restrictions, Trish refrained from visiting the hospital’s emergency department. Instead, Trish booked an appointment with her family doctor in two weeks’ time after her quarantine was finished. Over the next week, Trish complained of progressive muscle weakness in her hands and feet. She had difficulty grasping items with her hands and reported difficulty climbing stairs. During the subsequent week, Trish reported decreased coordination while ambulating and unusual shortness of breath and fatigue. Prior to the visit with her family doctor, Trish lost her balance and fell while walking upstairs, which led to admission to the hospital’s emergency department. Although no injuries were sustained by the fall, diagnostic investigation continued and Trish was diagnosed with acute motor axon neuropathy caused by Campylobacter jejuni, and was treated immediately with a high dose of intravenous immunoglobulin (IVIg) therapy[10]. Trish spent the next two weeks in the intensive care unit (ICU), where she required mechanical ventilation due to respiratory muscle weakness. The acute health care team performed passive range of motion, splinting, airway clearance techniques, and frequent repositioning to prevent the development of contractures, deep vein thrombosis, pressure ulcers, and other complications. Prior to discharge, Trish was educated by an interdisciplinary team (including a physiotherapist, occupational therapist) about the rehabilitation plan, safety considerations, and was also fitted for a 4-wheel rollator to assist in her recovery. It has been 12 weeks since her discharge from the hospital. No other medical conditions or comorbidities have developed, and her medical status has remained stable. The sub-acute phase of rehabilitation will be patient-specific, utilizing an ICF framework to address body function and structural impairments, activity limitations, and participation restrictions. In addition, health care providers will employ a safety-based approach to prevent patient exhaustion, and promote patient confidence in performing functional activities or tasks. Client Characteristics[edit | edit source] Patient Profile (PP): Trish Jones, 62 year old female Medical Diagnosis: Acute motor axonal neuropathy Nature of the Condition: Sub-acute phase of the disease, in recovery Primary Complaint: Weakness in muscles of hands, wrist, ankle, and feet, Fear of falling Decreased mobility and coordination Self-reported fatigue Past Medical History (PMHx) Rotator Cuff Surgery 10 years ago (fully healed and no associated functional impairments) Campylobacter jejuni infection 12 weeks ago Medications (Meds): High dose of intravenous immunoglobulin (IVIg) therapy as needed Primary Reason For Referral: Recovery from AMAN Inability to perform certain ADLs without assistance Mobility levels, balance, coordination, joint range of motion, muscle strength/endurance have not returned to pre-AMAN levels Decreased aerobic capacity contributing to feelings of fatigue Decreased participation in previously enjoyed leisure activities/hobbies and physical activities Examination Findings[edit | edit source] Subjective[edit | edit source] Presenting Condition:[edit | edit source] Trish reported that she was feeling unstable and did not have confidence when walking around. She stated that both her distal upper (hands, wrist) and lower extremities (ankle, feet) were weak, and that she was having difficulties performing basic activities such as opening a can, climbing stairs, or walking around her home. She has reported a loss of mobility due to muscle weakness in her ankles, which she further contributes to her instability when performing everyday tasks. Trish notes that she has enough hand and wrist strength to carry light objects but fatigues quickly. Trish is able to ambulate 15 minutes with assistance from a rollator walker and AFO (ankle foot orthotic), however she proceeds with a slow gait speed and wide base of support. She does not report any pain while walking, but mentions that she is quite fatigued after walking and needs around 2 hours of rest before being able to walk again. Trish notes that fatigue is worse in her lower extremities as opposed to her upper extremities, and predominantly affects her ankle and feet. Also, Trish reports extreme difficulty when ascending and descending stairs, needing assistance from her husband due to fear of falling, lack of balance, and motor weakness. In the assessment, Trish states she has been feeling anxious and sad since her diagnosis, and she is concerned that it may impact the quality of her relationship with her husband and daughter as she relies on their assistance. With help from an occupational therapist, her home is being prepared for modifications to better meet her current needs and presentation. Furthermore, Trish also sees a neurologist once a month to monitor the progress of her condition and medication. She lives in a 2 story home in the heart of the city. She has been having major difficulties navigating the stairs, and as a result her family helped her move her bedroom down to the first floor, instead of the second. She hopes to be able to climb 15 steps required to go to the second floor of her house. Social History[edit | edit source] Trish lives with her husband Carlos, while her daughter Mia lives two blocks away from her. Carlos and Mia have been supporting Trish since her diagnosis. Prior to her retirement, Trish worked as a Pilates instructor at the local YMCA, and had normally maintained high levels of physical activity before her diagnosis. Her husband is a retired Kinesiologist, who has been running weekly group circuit training sessions at the YMCA, in which Trish participated faithfully. While Trish is progressing slowly, she is concerned that she may never be able to participate in her previous enjoyable physical activity/exercise classes. Prior to her diagnosis, Trish enjoyed going on hikes with her family and friends, and participated in an adult bowling league which took place once per week. Since her diagnosis, Trish has not been able to hike or bowl with her friends, and reports feelings of isolation without them. Yellow flags:[edit | edit source] Score of 9 on the General Anxiety Disorder-7 (GAD-7) scale Reports feeling sad and anxious since her diagnosis Concerned that she may be placing increased strain on her family due to her increased reliance Does not believe that physiotherapy will help recover her ability to navigate stairs Reports feelings of isolation due to reduced participation in activities with friends and family Primary Goals:[edit | edit source] Trish’s main goal is to be able to ambulate independently or with assistance from a single point cane for at least one hour, so she can go back to exploring her community neighbourhood without the use of a rollator. She would also like to return to participating in her husband’s weekly group circuit training sessions, as she felt that it was a great form of physical activity and a great opportunity to bond with him. Finally, she would like to get back to bowling with friends in their adult community bowling league, as it is a huge source of social support for her. Utilizing physiotherapy, Trish is hoping to gain more confidence and balance when walking, be able to carry heavier objects such as grocery bags into her home, and open cans without assistance from her husband. Also, she would like to be able to navigate stairs with minimal assistance, however she is unsure if that is a realistic goal for her given her current condition. Objective[edit | edit source] Observation[edit | edit source] Client has bilateral motor weakness in her distal lower limbs (ankle, feet, toes) and bilateral motor weakness in her distal upper limbs (hands, fingers, wrist) Patient enters clinic using a rollator walker, patient has wide base of support when using rollator Bilateral foot drop Skin Inspection No signs of deep vein thrombosis such as swelling or calf pain. No pressure ulcers, cuts or bruises. Sensory Testing[edit | edit source] Normal sensation was reported with the following tests Tactile localization Light touch Proprioception and kinesthetic sense Stereognosis Visual field test Neurological Scan for Upper and Lower Body[edit | edit source] Dermatomes: normal sensation bilaterally in C1 to T1 dermatomes and L1 to S2 dermatomes. Myotomes: weakness in the following myotome groups: C6 wrist extension C7 wrist flexion C8 finger flexion T1 finger abduction L4 ankle dorsiflexion L5 big toe extension S1 ankle plantarflexion Reflexes Achilles reflex → no response (0), bilaterally Quadriceps tendon → weak (1+), bilaterally Biceps and triceps tendon reflexes → normal (2+), bilaterally Upper motor neuron reflexes (Babinski and Clonus) → normal, bilaterally Cardiorespiratory Assessment[edit | edit source] Table 1: Vital Signs and Ventilatory Muscle Strength measured at the time of initial assessment. Vital Sign Measure Interpretation Heart Rate (HR) 78 bpm WNL Blood Pressure 128/82 mmHg WNL Respiratory Rate 18 bpm WNL Ventilatory Muscle Testing Measure Interpretation Maximum expiratory pressure (MEP) 100 mmH2O WNL Minimum inspiratory pressure (MIP) 65 mmH2O WNL Function and Mobility Testing[edit | edit source] Grip strength: left hand 10 Kg, Right hand 11 Kg. Bed mobility: Trish is able to independently roll and sit up in bed without assistance. Transfers: Trish is able to transfer without assistance from chair to standing and walk short distances using a rollator and ankle foot orthotic Trish is unable to maintain balance in response to small perturbations so she is at risk of falls. Outcome Measures[edit | edit source] Table 2: Outcome measure scores at the time of initial assessment. Note: GBS= Guillain-Barre Syndrome; TUG= Timed Up And Go; BBS= Berg Balance Scale; 6-MWT= 6-Minute Walk Test. Outcome Measure Score Interpretation GBS disability score 3/6 Able to walk 5 meters with an aid TUG 40 seconds High risk of falls BBS 31/56 High risk of falls 6-MWT 237 meters Low function and mobility Range of Motion and Manual Muscle Testing (MMT) Table 3: Active range of motion (AROM), passive range of motion (PROM), and manual muscle testing (MMT) scores per joint at time of initial assessment. Joint AROM (degrees) L/R PROM (degrees) L/R Bilateral MMT score (out of 5) Metatarsophalangeal joint Unable to move (bilat) Flexion 30/30 Extension 80/80 1 flexion, 1 extension Subtalar Unable to move (bilat) Inversion 30/30 Eversion 10/10 1 inversion, 1 eversion Ankle Unable to move (bilat) Plantarflexion 45/49 Dorsiflexion 18/20 2- plantarflexion, 2- dorsiflexion Knee Flexion: 110/ 115 extension: 10/ 8 Within normal limits (WNL) 4- flexion, 4- extension Hip Flexion: 100/ 105 Extension: 10/ 10 External rotation: 25/ 40 Internal rotation: 20/ 38 Abduction: 35/ 38 Adduction: 20/ 20 WNL 4- flexion, 4- abduction Shoulder Flexion: 90/ 115 Abduction: 85/ 100 External rotation: 50/ 70 Internal rotation: 55/ 75 WNL 4+ flexion, 4 abduction Elbow Flexion: 145/ 147 Extension: 0/0 WNL 4+ flexion, 4+ extension Wrist Flexion: 60/ 62 Extension: 50/ 54 WNL 4- flexion, 4- extension Metacarpophalangeal joint Flexion: 60/ 62 Extension: 10/ 10 WNL 3+ flexion, 3+ extension Clinical Impression[edit | edit source] Physiotherapy Diagnosis[edit | edit source] Trish’s primary clinical presentation is bilateral peripheral motor weakness without sensory loss which was due to a medical diagnosis of Acute Motor Axonal Neuropathy (AMAN), a variant of Guillain-Barre Syndrome. Her symptoms are more severe in the lower extremities, where she presents with loss of ROM and weakness in the dorsiflexors and plantarflexors. Consequently, she exhibits decreased ability to maintain balance, an increased fear of falls, and is also unable to ambulate without a mobility aid (4-wheeled rollator). Trish does does not require any ventilatory support, however she experiences extreme fatigue after 15 minutes of ambulation that subsides with rest. Although retaining full ROM in the upper extremity, she has decreased grip strength which limits her ability to perform certain ADLs independently such as turning door knobs and opening cans. Currently, Trish is unable to participate in activities she previously enjoyed including pilates, bowling nights with her friends, and attending her husband’s fitness classes. Problem List (based on the ICF framework)[edit | edit source] Body Function and Body Structure Impairments Inability to perform full AROM ankle dorsiflexion or plantarflexion bilaterally Motor weakness in peripheral joints (wrist, ankle, and interphalangeal joints) Fatigue after walking 15 minutes Poor balance and coordination during standing and gait Activity Limitations Limited ability to perform grasping actions (open jars, turn doorknobs, carrying groceries) Inability to go up stairs without 2 railings due to fear of falls Requires rollator for ambulation Participation Restrictions Unable to participate in husbands group circuit training classes Unable to participate in hikes with family and friends since her diagnosis Inability to participate in weekly adult league bowling nights Anxious to interact with friends as she is embarrassed about her compensated ambulation patterns Intervention[edit | edit source] The purpose of the listed interventions are to: Help patients to maintain and regain muscle strength, ROM, and function during the remyelination process. It is important to note that although therapy does not directly facilitate nerve tissue repair, it allows patients to optimally use the muscles during the healing process. Creating a patient-centered treatment plan following the ICF framework to address body function and structure impairments, activity limitations, and participation restriction Educate patients on the proper use of mobility aids and functional adaptations that allow patients to resume activities as close to their previous level of activity. Will use a safety-based approach to help patients recognize and perform activities in ways they feel safe to do so. Patient-Specific Goals[edit | edit source] During the next 2 weeks, Trish will perform daily active ankle range-of-motion exercises using visualization techniques and a belt to self-facilitate movement. By week 3, Trish will be able to attend and participate in a seated, modified version of her husband's weekly group circuit training. Within 4 weeks, Trish will be able to ambulate for 20 minutes with a rollator without experiencing fatigue that lasts more than 15 minutes (fatigue is defined in this context as lack of energy that prevents her from performing/return to daily activities). Management Program during Recovery Phase[edit | edit source] General notes for exercises[11]: Exercises should be matched to patients activity, performed in short intervals and not overly fatiguing. Exercise progression should only occur when patient shows improvement in exercises for a week, without deterioration of status (ie. increased fatigue) Table 4: Detailed Explanation of Physiotherapy Interventions Intervention Rationale FITT (frequency, intensity, time, type) principles Additional Notes Education: Proper postural education (neutral spine, sitting up tall, shoulders back) and management (recommend postural adjustments that will help her maintain proper posture - lumbar roll) Safety consideration when using a gait aid Understand disease progression, prognosis, patient-specific goals, and treatment plan Role of caregiver (supportive and motivating but should encourage patient self-managment) Indicators to decrease or increase the level of intervention, and safety associated with each exercise Having the patient understand the development and progress of their condition can increase patient buy-in, increases patient safety, and allows patients to take a larger role in their own rehabilitation. Education should begin during the first session of therapy. Use of pamphlets or videos may be helpful too. As therapists, it is important to encourage patients without making promises on patient’s recovery time and degree of recovery[12]. Range-of-motion: Neuro-proprioception techniques to facilitate ankle dorsiflexion and plantarflexion AAROM and PROM techniques that facilitate ankle ROM, to keep existing neuro-muscular junctions active, to encourage motor control. 10 reps x 3 times/day, performed daily Even if muscles are weak and full ROM cannot be achieved independently, the patient is encouraged to visualize the action during the exercise Since sensation is not lost in these patients, can use external cues on on leg to promote muscle contraction PROM can be assisted by the therapist AAROM can be done as part of the home intervention plan using a belt to assist with ankle dorsiflexion PROM technique can be done in rehabilitation or by a caregiver in a home setting. Muscle strengthening: Examples include: supine glute bridges seated hip flexion mini-squats with hands supported on counter/ stable surface theraband-resisted shoulder flexion and abduction bicep curls Continue to strengthen and maintain larger lower (ie. quadriceps glutes, and hamstring) and upper extremities (ie. biceps and tricep) and trunk prime movers, as these are required to regain previous activity levels and function 8 reps x 3 sets with adequate rest in between (patient dependent) 3x/wk Intensity: low to moderate level of exertion Type: isometric and isotonic resistance training Patient should be aware of fatigue levels using the Rate of Perceived Exertion on the Borg Scale (begin at low intensity and progress to moderate intensity) Multi-joint exercises can also be included as it encourages muscle synergies (coordinate prime mover and stabilizer action)[13] Should be taught in rehabilitation and performed in home setting. Grip Strengthening: Hand therapy:GRASP (graded repetitive arm supplementary program[14][15] This program focuses on upper extremity strengthening, fine motor tasks and repetitive functional tasks. Although this program is only validated in the acute stroke population, this can be beneficial if adapted to patients ability to improve grip strength and general upper functional outcomes. 1 hour of exercises, daily 8 weeks Remember to modify and adapt exercise to patient-specific fatigue levels, allow for rest as necessary GRASP is a “homework-based” program that can be performed at home. Should be done in home setting. Cardiovascular aerobic training: Examples Walking using gait aid Arm Cycle ergometer Stationary bike Safe, low-level, aerobic exercise that does not go past level of fatigue is indicated for positive clinical outcomes 30 min total (can be broken up into 5-10 minute bouts) 3x/week Recommended to do walking with 4-wheeled rollator as this is a most functional activity Patient should be aware of fatigue levels using the Rate of Perceived Exertion on the Borg Scale (begin at low intensity and progress to moderate intensity)[16] Can be done in rehabilitation or home setting. Balance and Gait Parallel bar gait training (walking, weight shift, challenge balance) Body-weight supported treadmill training[17] Educate proper and safe use of gait aid Training balance can help improve patient’s confidence in functional activities that require balance and help patients improve motor control patterns Always perform this in a safe setting and with a therapist spotting the patient. Can be performed in a home setting if caregiver is taught the proper way to support patient. Use of ankle-foot orthosis as indicated Use gait belt for safety as indicated By using a body weight treadmill, the patient may feel safer and more comfortable to ambulate without holding on to a support as they are supported by a harness. External facilitation to encourage ankle dorsiflexion and plantarflexion as indicated. Parallel bar and body-weight treadmill can be done in rehabilitation setting. Functional Training Step up Sit to stand Reaching for objects while standing Adapted pilates (e.g seated pilates) Putting it all together: Functional training takes a multifaceted approach to help patients strengthen, improve ROM, coordination and balance in a patient relevant 10 reps (or as many as tolerated), 2x/day preformed daily For pilates perform 1-2x/wk When these exercises are performed at home, make sure that they are performed safely and within the patient’s limit to prevent excessive fatigue Patient should be aware of fatigue levels using the Rate of Perceived Exertion on the Borg Scale (begin at low intensity and progress to moderate intensity)[16] Can be done in rehabilitation or home setting. [15] [17] Outcome[edit | edit source] After 12 weeks of physiotherapy sessions, Trish has improved significantly on multiple components of her diagnosis that have impacted her independence the most, such as her strength, range of motion, balance, and gait. She is also experiencing less fatigue in comparison to her initial assessment and can successfully ambulate over 25 minutes before feeling mild fatigue. Trish was assessed at various intervals throughout her treatment to ensure the interventions matched and challenged her function and activity levels. Trish’s progression at the 6-week interval is as follows: Re-assessment 6-Weeks after Initial Assessment[edit | edit source] Table 5: Outcome Measures at 6-Weeks Re-assessment Outcome Measure Score Interpretation GBS disability score 2/6 Able to walk for 5m or more without an aid TUG 33 seconds High risk of falls; requires gait aid BBS 40/56 Increased functional balance 6-MWT 307 meters Increased distance; improvement in patient’s mobility Table 6. Range of motion (ROM) and Manual Muscle Testing (MMT) at 6-Weeks End of Week 6 Update Joint AROM (degrees) L/R PROM (degrees) L/R Bilateral MMT score (out of 5) Metatarsophalangeal joint Flexion: 10/8 Extension: 15/12 Flexion: 33/32 Extension:84/84 2+ flexion, 2+ extension Subtalar Inversion: 7/10 Eversion: 4/4 Inversion: 34/34 Eversion: 14/16 3 inversion, 3 eversion Ankle Plantarflexion: 13/ 15 Dorsiflexion: 8/7 Plantarflexion: 47/51 Dorsiflexion: 22/24 3+ plantarflexion, 3+ dorsiflexion Knee Flexion: 117/ 120 Extension: 6/6 WNL 4 flexion, 4 extension Hip Flexion: 110/113 Extension: 13/14 External rotation: 30/42 Internal rotation: 25/42 Abduction: 39/41 Adduction: 25/25 WNL 4 flexion, 4 abduction Shoulder Flexion: 110/ 123 Abduction: 100/ 104 External rotation: 57/74 Internal Rotation: 64/79 WNL 4+ flexion, 4+ abduction Elbow Flexion: 153/153 Extension: 0/0 WNL 4+ flexion, 4+ extension Wrist Flexion: 66/69 Extension: 57/62 WNL 4 flexion, 4 extension Metacarpophalangeal joint Flexion: 65/68 Extension: 15/16 WNL 4 flexion, 4 extension Progression of her scores can be attributed to intervention education provided to Trish. She is eager to participate in her rehabilitation (strength, range of motion, and balance interventions) as she believes it will help her return to previous levels of independence and participate in her weekly hobbies. As Table 5. illustrates, Trish’s TUG score has improved significantly (from 40 seconds to 33 seconds, MCID is 3.4 seconds[18]). She also improved in her BBS score and has significantly increased in her 6-MWT (MCID estimated at 54-80 meters[19]). These improvements can be attributed to the parallel bar and body-weight treadmill gait training that was implemented. Trish reports that she sees increases in proficiency as she performs the prescribed muscle strengthening and functional exercises and believes she can return to her previous lifestyle. Trish also reported feeling decreased fatigue after physiotherapy each session. Trish is still advised to continue practicing neuro-proprioception techniques along with imagery to facilitate more ankle dorsiflexion and plantarflexion as part of the home intervention plan. Lastly, Trish reported feeling depressed at the very beginning of her treatment plan. We referred her to a psychologist at this stage to provide psychological support for her and her family and gain motivation to participate in her plan of care[20]. Since then, she reports improvement and is looking forward to advancing to the next stages of her treatment plan. Re-assessment 12-Weeks after Initial Assessment[edit | edit source] At the end of week 12, a final assessment was taken again. Her results are as follows: Table 7: Outcome Measures at 12-weeks Re-Assessment Outcome Measure Score Interpretation GBS disability score 2/6 Able to walk for 5m or more without an aid TUG 27 seconds Patient still requires gait aid BBS 46/56 Decreased risk of falling; functional balance improved 6-MWT 382 meters Progression in distance; increase in patient’s mobility Table 8. Range of motion (ROM) and Manual Muscle Testing (MMT) at 12-Weeks End of Week 12 Joint AROM (degrees) L/R PROM (degrees) L/R Bilateral MMT score (out of 5) Metatarsophalangeal joint Flexion: 13 /12 Extension: 20/19 Flexion: 35/35 Extension: 86/88 3 flexion, 3 extension Subtalar Inversion: 14 /14 Eversion: 8/9 Inversion: 35/35 Eversion: 16/17 4 inversion, 4 eversion Ankle Plantarflexion: 21/ 25 Dorsiflexion: 11/14 Plantarflexion: 49/52 Dorsiflexion: 24/25 4- plantarflexion, 4- dorsiflexion Knee Flexion: 126/132 Extension: 3/3 WNL 4+ flexion, 4+ extension Hip Flexion: 117/120 Extension: 16/18 External rotation: 35/45 Internal rotation: 32/45 Abduction: 44/46 Adduction: 30/31 WNL 4+ flexion, 4+ abduction Shoulder Flexion: 105/122 Abduction 120/122 External rotation: 65/80 Internal Rotation: 70/82 WNL 4+ flexion, 4+ abduction Elbow Flexion: 157/158 Extension: 0/0 WNL 4+ flexion, 4+ extension Wrist Flexion: 70/70 Extension: 64/65 WNL 4+ flexion, 4+ extension Metacarpophalangeal joint Flexion: 72/73 Extension: 20/22 WNL 4+ flexion, 4 extension With the help of the body-weight treadmill, Trish is able to practice ambulation and train proper gait motor control in a supported environment. Along with this intervention, she reports increased ankle strength which has allowed her to ambulate more confidently. Trish also has a significantly higher TUG score (from 33 seconds to 27 seconds, MCID is 3.4 seconds[18]). She also gained significant improvements in her BBS overall and significantly improved her walking endurance as shown in the 6-MWT score (MCID 54-80 meters[19]). As illustrated above in Table 8, Trish has overall increased strength and has almost achieved full ROM in her ankle joint. She reports feeling reduced fatigue after physiotherapy sessions in her daily walks and can walk for up to 30 minutes before experiencing fatigue. Her grip strength also increased, which is reflected in her ability to perform ADLs independently. Lastly, Trish reported feeling positive about her progression in therapy and mentioned feeling more independent when participating in her individual and social activities. Discharge[edit | edit source] Based on the current status of Trish’s progress, we recommend that Trish decrease the frequency of her appointments from weekly sessions to monthly check-ups. In these sessions we will reassess her progress and help her work towards her goal to progress from a 4-wheeled rollator to a quad-cane, if indicated. Together with an occupational therapist, Trish will find ways to modify her home environment based on her needs[21]. We also recommended that Trish take part in aquatic therapy programs, as it is beneficial for patients with neurological conditions, such as AMAN[22]. Aquatic physical therapy can result in reduced pain, further reduce joint stiffness, promote greater physical function, and increase the quality of life for our patient[23]. We will have ongoing discussions with Trish as well as the interprofessional health team involved in her health. [24] Interdisciplinary Team[edit | edit source] Patients with a diagnosis of Acute Motor Axonal Neuropathy (AMAN) require holistic care from a variety of healthcare professionals in order to help optimize patient outcomes. Patients with AMAN can experience lasting motor weakness deficits, which require interdisciplinary rehabilitation approaches to support and facilitate patients in regaining their previous level of functioning and independence. It is important to involve patients and/or family caregivers in their unique care, the decision-making process, as it promotes autonomy and facilitates self-efficacy. A multidisciplinary healthcare team is needed to effectively manage and treat patients with a diagnosis of AMAN. We have listed different healthcare professionals involved in the treatment and management of patients with AMAN along with their roles. Note, this list is not exhaustive. Physiotherapist: Assess, monitor, and prescribe interventions to improve patients respiratory muscle strength/functioning, airway clearance techniques Assess, monitor, and prescribe interventions to improve patients posture, range of motion, strength, endurance, balance, functional independence in performing ADLs Facilitate bed mobility and frequent re-positioning in the acute stages of the disease to prevent the development of pressure ulcers and deep vein thrombosis Utilizing pain management strategies and incorporating the biopsychosocial model Educating, fitting, prescribing patients with mobility aids to facilitate functional independence and reduce the risk of falls [25] Occupational Therapists Focus on muscle strengthening and help patients relearn movement strategies to improve performance of ADLs with minimal assistance Teach patients new techniques or modified techniques to facilitate functional independence Find ways to modify the patient's home environment based on patient limitations or patient-specific needs (ie. moving her bed to a first floor room before she is able to walk up stairs)[21] Psychologists Provide psychological support for patients and their families Upon diagnosis of AMAN, patients can experience anxiety, depression, or reduced motivation. Therefore, regular psychological counselling is important to help patients assess, monitor, and manage their newly-experience emotions associated with their diagnosis In addition, there are support groups for patients with AMAN, which provides an outlet for patients to recognize they are not alone and that there is a group for them to talk to which can help minimize social isolation [20] Neurologist Role in diagnosis and treatment of AMAN Neurologists can perform diagnostic procedures to aid in the diagnosis of AMAN In addition, neurologists can prescribe medications in the treatment of AMAN, and play an important role in monitoring patient progress[26] Discussion[edit | edit source] Acute Motor Axonal Neuropathy (AMAN) is a variant type of Guillain-Barre syndrome, characterized by acute progressive motor weakness, areflexia, ataxia, oculomotor dysfunction and absence of sensory symptoms[1]. Prognosis for this condition is typically positive, with severe cases continuing to recover movement and function up to four years after their initial diagnosis[3]. Deficits observed in motor function, range of motion, aerobic and muscular endurance, and muscle strength are key factors that continue to limit the quality of life of individuals suffering from AMAN[26]. In her assessment, Trish presented with many of the listed signs and symptoms, which we measured via outcome measures in accordance with the ICF (such as the GBS disability score and TUG). The outcome measures were used to quantify her initial level of functioning, and as comparable signs to monitor her physiotherapy progress (at 6- and 12- weeks). Initially, Trish’s signs and symptoms led to a decrease in her level of independence and her ability to participate in enjoyable recreational activities such as pilates or bowling, leading in a reduction in her overall quality of life and mental health. Emphasis should be placed on the favourable prognosis of this condition when educating patients, along with highlighting the importance of physical therapy and a treatment approach revolving around functional activity. Along with patient-stated goals, Trish’s 12-week treatment plan was structured with the ICF framework in mind. Her treatment focused on ROM, muscle strengthening, meaningful functional activities, aerobic exercise, and balance training to reduce her risk of falls. Throughout her engagement with the intervention program Trish demonstrated excellent progress and has become more functionally independent. An increase in her ankle strength (MMT testing) has allowed her to ambulate more confidently, and her ability to effectively counter light external perturbations has reduced her fear of falls. Trish still ambulates around her home and community with a rollator walker, although with notably improved endurance, and has not yet been able to get back to bowling with her friends. Her desire to progress to a cane and return to her husband's workout classes make her an excellent candidate to continue with rehabilitation. Physiotherapy continues to be beneficial as it can provide Trish with more education and functional-focused intervention that can help her achieve her goals. Trish’s previously high levels of physical activity, motivation to return to activity, and continued support from her family are strong indicators that she will continue progressing, ideally to a level where she does not feel limited by her AMAN diagnosis. AMAN patients and those with similar conditions continue to benefit from the expertise of a multidisciplinary team well beyond their diagnosis, to ensure a maximization of their quality of life. Self Study Questions[edit | edit source] 1. Which of the following symptoms is NOT typical of patients with AMAN A) Bilateral motor weakness B) Graphesthesia C) Loss of balance D) Increased fatigue 2. The main characteristic differentiating acute motor axonal neuropathy (AMAN) from Guillain-Barre Syndrome (GBS) is A) GBS patients may experience a decrease in respiratory function while AMAN patients do not B) AMAN patients may experience sensory deficits while GBS patients do not C) AMAN involves purely motor deficits D) Prognosis for AMAN is poor while GBS prognosis is typically positive 3. The most effective treatment approach for AMAN focuses on (choose the best answer) A) Focus on functional activity B) Sensory training C) Muscle and endurance training D) Aerobic exercise E) A,C, and D only 4. Which of the following is NOT a proposed mechanism of recovery in AMAN? A) Terminal motor nerve axonal regeneration B) Collateral reinnervation C) AgglutinationGuillain-Barré Case Study: David - PhysiopediaAbstract This case study follows the case of David Atkin, who was diagnosed with Guillain-Barré Syndrome (GBS) following a bacterial gastro-intestinal infection of Campylobacter Jejuni. GBS is an autoimmune disease characterized by bilateral progressive motor deficits beginning in the hands or feet. David had progressive loss of motor control and sensation primarily to the feet which led him to checking into the emergency department. Rapid progression of his symptoms occurred, affecting his respiratory muscles and leading to respiratory failure. David consequently had to be intubated and placed on a mechanical ventilator. Following the acute phase, the intervention plan focuses on aspects of mobility, strength, aerobic capacity, and balance to achieve his goals related to body structure, activity, and participation. This case provides a typical case presentation of GBS, in addition to possible assessment methods, outcome measures, and technological uses to treat this disease. Introduction[edit | edit source] Guillain-Barré Syndrome (GBS) is a rare autoimmune disease that affects the peripheral nervous system (PNS). The exact mechanism is unclear, but the majority of GBS cases are triggered following bacterial or viral infection[1]. GBS can also be triggered by trauma, surgery, cancer, or vaccination[2][3]. GBS has 3 subtypes, however the most common subtype is acute inflammatory demyelinating polyradiculoneuropathy, caused by an autoimmune reaction where the immune system targets and breaks down the myelin sheath surrounding peripheral nerves, therefore a disease of demyelination[4]. The first symptoms noticed are typically numbness, tingling, or pain (alone or in combination) beginning in the feet and ascending proximally towards the head. Over the course of days to weeks, there is onset of progressive muscle weakness in the extremities and potential paralysis[5]. Guillain-Barré Syndrome can also lead to weakening of the respiratory muscles and eventual respiratory failure[1]. Many complications can arise during the acute stage of GBS including: deep vein thrombosis, pulmonary embolism, heart attack, pneumonia, and infection. Death occurs in about 2.6% of all cases of GBS[6]. Signs and Symptoms[7][8][9][edit | edit source] Bilateral numbness, tingling, or pain (alone or in combination) that begins in the hands and feet Progressive bilateral weakness of the extremities Impaired gait and balance Weakness of facial muscles Difficulty with swallowing or speaking Double vision Severe pain that may worsen during the night Changes to bowel/bladder control Paralysis Respiratory failure Autonomic dysfunction (abnormal changes to heart rate and blood pressure) Epidemiology[edit | edit source] More common in men[10] Risk increases with age[10] 3,000 to 6,000 cases per year in the United States[11] Incidence of subtypes varies between countries[2] Subtypes[edit | edit source] Various symptom presentations of GBS [12] There are several subtypes of GBS, each presenting differently and affecting different populations. In North America, the most common form of GBS is Acute Inflammatory Demyelinating Polyradiculoneuropathy (AIDP)[2]. AIDP is characterized by the presence of sensory symptoms, muscle weakness, involvement of cranial nerves, and autonomic dysregulation. The focus of this case study is on the AIDP subtype; the presentation follows the classic sensorimotor pattern of symptoms. Prognosis[edit | edit source] The GBS Disability Scale is used to measure the level of symptoms at milestones throughout recovery[13] [1] Symptom severity can vary and as such, the degree of recovery and the timeline also varies. Many patients have chronic symptoms and changes to their functional status following their bout with GBS. The Guillain-Barré Syndrome (GBS) Disability Scale is used to measure the degree of recovery[14]. Recovery can range from full recovery for some patients to significant functional impairment for others. The prognosis of GBS depends primarily on age of the patient and on the severity of symptoms two weeks after onset. Older individuals are more likely to have lasting effects following an episode of GBS. The course of GBS includes progression of symptoms of days to weeks before reaching a plateau period where symptoms stabilize. The plateau period typically lasts weeks but in some cases may last months. Following that there is a gradual recovery where function returns, however, this can take months and many are left with some level of disability.[9] Roughly 30% of GBS cases will lead to weakness of the respiratory muscles which may eventually lead to respiratory failure[15]. If this occurs, the patient will have to be intubated and placed on mechanical ventilation. Mechanical ventilation can lead to complications such as pneumonia which can affect negatively affect recovery. Client Characteristics[edit | edit source] Patient Profile[edit | edit source] Initials: D. A (David Atkin) Preferred Name: Dave Age: 35 years DOB: 12 June 1987 Gender: Male Height: 172cm Weight: 90kgs Significant Presentation: Sudden onset of symmetrical bilateral acroparesthesia and paralysis in lower extremities. Patient presented with areflexia and flaccidity bilaterally on testing. History of Present Illness[edit | edit source] Date of Admission: 30th March 2023 Type of Admission: Self- referral. Patient woke up and was unable to move his lower limbs. Patient brought in an ambulance to Kingston General hospital (KGH). Initial Diagnosis: Acute inflammatory demyelinating polyneuropathy (AIDP) a sub-type of Guillain-Barré syndrome (GBS). The patient was admitted in KGH on 14th March 2023 with a gastrointestinal infection (caused by Campylobacter Jejuni[1]) which is one of the significant diagnostic indicators of GBS. Date of Onset: 30th March 2023 Treatment to Date (20th April 2023): Intravenous immunoglobulin therapy and plasmapheresis, the previous PT and PTA worked on bed mobility. Present Status: Alert and oriented. Minimal regain of motor and sensory loss bilaterally on both extremities. (Note: On 15th April 2023, patient was transferred to ICU and intubated after acute bradycardia, bilateral facial paralysis and acute respiratory distress. Patient had shown progressive bilateral symmetrical sensory and motor loss in both upper and lower extremities.) Precautions / Contraindications: Patient may exhibit occasional orthostatic hypotension if they are assisted out of recumbent position. Past Medical History[edit | edit source] Allergies: Peanuts and tree nuts Medication[edit | edit source] Venus thromboembolic prophylactics to prevent Deep Vein Thrombosis and Pulmonary Embolism Amlodipine 5mg per day for hypertension Health Habits[edit | edit source] Smokes one pack of cigarettes every day since the age of 30 years (5 pack-years), and reports drinking 1 beer (3 times a week) Social History[edit | edit source] Works as a manager at a software development firm in Kingston The patient lives in a 2-story independent house that has 12 stairs with his wife and 2 daughters (aged 9 and 6) Functional History[edit | edit source] Resistance training at GoodLife fitness twice a week. Was independent with BADLs and IADLs Fell once while skateboarding and fractured his left wrist in December 2019 Current Functional Status[edit | edit source] Has independent bed mobility Can maintain seated posture alone but needs assist x1 to transition from laying to sitting and assist x2 to stand Unable to ambulate due to balance impairments combined weakness in extremities Examination Findings[edit | edit source] Observation[edit | edit source] The patient is alert and oriented - no lines or tubes present; currently lying in semi-reclined position. Patient appears to be in good spirits and expressed optimism for his recovery. Mobility[edit | edit source] Bed mobility: independent to slide up, down, sideways, roll onto side Lie to sit: minimal Ax1 Sit-to-stand: unable to stand on own requires max Ax2 Transfers: moderate Ax2 required for pivot transfer Personal Care/ADLs[edit | edit source] Assistance needed for dressing and bathing Currently utilizing a urinary catheter Independent to feed oneself Gait[edit | edit source] Currently unable to ambulate due to lower extremity weakness Need fitting for gait aid when required Range of Motion[edit | edit source] Muscle group AROM PROM Muscle Group AROM PROM All shoulder movements WNL WNL Hip flexion 45 WNL All elbow movements WNL WNL Hip extension 10 WNL All wrist movements WNL WNL Hip adduction WNL WNL Hip abduction WNL WNL Knee extension WNL WNL Knee flexion 100 WNL Dorsiflexion WNL WNL Plantar flexion WNL WNL Muscle Strength[edit | edit source] Muscle group Right Left Muscle Group Right Left All shoulder movements 4 4 Hip flexion 3- 3- Elbow flexion 4- 4- Hip extension 3- 3- Elbow extension 4 4 Hip adduction 4 4 Wrist flexion 4- 4- Hip abduction 3 3 Wrist extension 4- 4- Knee extension 3+ 3+ Knee flexion 3- 3- Dorsiflexion 3+ 3+ Plantar flexion 3+ 3+ Balance[edit | edit source] Seated: patient can maintain a seated posture independently once positioned, with SBA Standing: unable to independently stand, requires max Ax2 Common Comorbidities[16][edit | edit source] Hypertension Diabetes Hyperlipidemia Stroke Lung Infection Outcome Measures[edit | edit source] Functional Independence Measure (FIM) The FIM is a common outcome measure used to determine the ability to perform activities of daily living. It is made up of 18 individual items consisting of motor and cognitive functioning, that are scored from 1 (total assistance) to 7 (independent). Each item is added together to get an overall level of independence between 18-1126. The FIM is considered time-consuming but an easy-to-use, valid and reliable method that can be trusted in clinical settings to assess patients with various conditions.[17] 10-Meter Walk Test The 10-meter walk test is a useful tool to measure gait speed and functional mobility. The tool uses the amount of time a patient takes to walk 10 meters, to provide a speed in m/sec. This can be compared to normative data to determine risk of falls and if a gait aid is indicated. The 10-meter walk test is a common tool used by clinicians and is seen as reliable and valid for a number of neurological conditions[18]. Medical Research Council Sumscore The Medical Research Council Sumscore is a common tool used to measure muscle strength. This tool considers six main muscle groups (bilaterally) and assesses their strength using a scale of 0-5. These six scores can then be added together to create a total sum ranging from 0 to 60[19]. It has shown to be a clinically useful tool for physiotherapists for neurological conditions. This tool has high intra-rate reliability, indicating it needs to be repeated by the same physiotherapist to assess change[20]. Clinical Hypothesis[edit | edit source] Analysis Statement 35 y/o male admitted to Kingston General Hospital (KGH) via ambulance on March 30th, 2023, due to inability to move lower limbs after waking up. Pt presents with bilateral acroparesthesia and paralysis in lower extremities, with bilateral areflexia and flaccidity; diagnosed with acute inflammatory demyelinating polyneuropathy (AIDP) - a sub-type of Guillain-Barre syndrome. Pt currently unable to ambulate due to impaired strength, sensation, and balance in lower extremities. This patient's prognosis tends to be positive with a variety of factors that favour his recovery including young age, previously being active, and a relatively high MRC Sumscore on initial assessment. However, the patient has several negative factors affecting his prognosis as well, these include having to take care of two daughters, being a current smoker, and an inability to ambulate. This patient would benefit from physiotherapy treatment focused on strengthening the lower extremities, improving balance, and increasing mobility. Problem List[edit | edit source] Unable to care for two young daughters (participation in role as a father) Inability to ambulate (activity) Lower extremity weakness in all major muscle groups (body function) Goals[edit | edit source] Long-Term Goals To be able to pick up and hold daughters safely within the next 4 months To be able to walk independently with a 2WW to get to the bathroom within the next 3 months Gain the lower extremity strength to be able to perform 10 arm-supported squats within the next 2 months Short-Term Goals To be able to maintain independently sitting balance for 10 minutes while reading and interacting with daughters in the next week Achieve a sit-to-stand and be able to stand with minimal support and a 2WW within the next 3 weeks. To be able to complete 10 repetitions of in-bed resistance exercises (glute bridge, quad-over-roll), twice a day within the next 2 weeks Intervention[edit | edit source] Treatment Plan Though there is no known cure to Guillain-Barre syndrome, physiotherapists play an important role in the functional recovery of GBS patients. In the acute phase of recovery of GBS, the patient likely will not tolerate active movement due to a rapidly worsening condition. Thus, at this stage the physiotherapist may play more of an advisory role, educating the patient on the prevention of deep vein thrombosis (DVTs), pressure sores, and contractures [21]. However, 90% of GBS patients reach clinical nadir within 4 weeks, with aspects of functional recovery occurring thereafter [22]. This is where the primary focus of our intervention plan in David’s case. General goals of physical therapy treatment for GBS patients include optimal muscle use at tolerable pain levels, in addition to the use of supportive equipment to recover function to pre-illness levels (as much as possible) [21]. Various exercise programmes have been shown to improve physical outcomes in patients with GBS, such as functional mobility, cardiopulmonary function, isokinetic muscle strength, and reduced fatigue[23]. We are recommending David complete PT treatment anywhere from 3-5x per week for 1 hr, as a higher intensity program relative to lower intensity program may lead to significantly reduced disability in GBS patients [24]. However, it is crucial to note that exercise should not be done to fatigue in patients with GBS, as this may cause central fatigue, loss of strength, and delay recovery[21]. Lastly, considering pain is a common symptom of GBS and can be prevalent throughout rehabilitation, strategies such as transcutaneous electrical nerve stimulation (TENS), heat, and sensory desensitization techniques may be of aid in alleviating pain and improving tolerance to exercise[21]. However, the unique pain experiences of individuals with GBS leads to varied outcomes when using these techniques. To achieve David’s short- and long-term goals, treatment will focus on mobility, range of motion (ROM), strength, balance, and cardiorespiratory endurance. As a secondary benefit, these multiple types of activities help facilitate sensory stimulation and desensitization techniques, along with using fine and gross motor skills for movement re-education and pain reduction [21]. Along with the justification of each physical outcome as part of our program, we have included specific exercises with FITT (frequency, intensity, time, type) parameters as a possible course of treatment: ROM To prevent muscle atrophy and other negative effects of disuse, it is important to help David get moving as soon as he is able [21]. A natural progression of range of motion exercises begins with passive range of motion, following by active assisted range of motion (AAROM), then finally to active movements. Equipment such as a powder board or a hydrotherapy pool can facilitate movement while limiting work on anti-gravity muscles [21]. Additionally, David may use proprioceptive neuromuscular facilitation (PNF) stretching to improve motor function and motor control [25]. Possible stretches David can do is a supine hip flexor stretch, supine figure 4 stretch, calf stretch, hamstring stretch, pec stretch, and elbow flexor stretch. These may be assisted by the physiotherapist, or the patient may use equipment such as a strap. Importantly, these stretches should not cause fatigue. Listed below are the FITT parameters to follow: Frequency 3x/week (or daily) Intensity slight discomfort, but no pain Type static stretching, PNF stretching Time 30-60s Strengthening When beginning a strength program with GBS, it is important to start slowly, with zero resistance and the inclusion of frequent rest breaks. Training should not be completed to fatigue; complaints of fatigue should not persist for >12-24 hours post session [21]. Strength training of the lower and upper extremity may increase functionality and quality of life in individuals with GBS [26]. In David’s case, strengthening should target the weakness observed in his lower extremities. Bed exercises may include isometrics (e.g., glute squeeze, quad squeeze, etc.), isotonic exercises (quad over roll, heel slides, glute bridges), and resistance band training (e.g., bicep curl, triceps extension, etc.). Once David achieves ambulation, strength training can progress to more functional movements for daily tasks (such as sit to stands and squatting) and achieving his personal goals (such as playing with his daughters). Frequqncy 3x per week (daily if tolerated to promote movement) Intesity Never to fatigue Type Isokinetic and isometric Time Variable Aerobic Capacity In addition to ROM and strengthening, other activities (such as aerobic capacity) can be beneficial for cardiovascular health and general conditioning [25]. The exercises vary, but may include an arm ergometer, cycling ergometer, walking. Cycling has been shown to elicit significant functional improvements in GBS patients [23]. Cardiorespiratory endurance will positively affect physical fitness and reduced fatigue, positively affecting all of David’s goals [25]. Frequency 3x/week Intensity Roughly 65% MHR or not exceeding 45% heart rate reserve[27]; 0 Watts- may choose to increase wattage as one progresses; Moderate RPE on BORG scale [21] Type Variable - Arm ergometer, cycling, walking Time 5-10 mins, up to 30 mins Balance Balance is an integral part of everyday life for GBS patients and thus should be included within meaningful activities. David can practice balance with sitting exercises, such as reaching for objects like the TV remote or a glass of water across the table. Standing balance exercises may be out of reach currently, but David can progress to these in the future. In due time, these may include trying to change clothes, getting in and out of the shower/tub, and re-introduction to leisure activities with minimal to no assistance. Along with other physically demanding activities, it is paramount with balance (both sitting and standing) to ensure adequate rest breaks are taken to reduce the risk of falling [21]. Frequency 3x/week (daily if tolerated) Intensity Never to fatigue Type Sitting and Standing balance exercises Time Sitting: 30-60 seconds as tolerated. Standing: 5-30 seconds as tolerated. Transfers (Bed Mobility) Transfers may be taught to maximize independence, help achieve David’s meaningful activities and ADLs, and prevent pressure sores in patients with GBS, particularly in the non-ambulatory phase of the condition. Some useful transfers to teach David include: Lying to sitting Sitting to standing Pivot transfer Frequency 3x/day (if tolerated) Intensity Never to fatigue Type Lying to sitting, sitting to standing, and pivot transfer Time Throughout the day as necessary Innovative Technology: Virtual Motor Rehabilitation System A key clinical feature of GBS is motor disturbances and muscle weakness that can become persistent and progressive. There has been evidence that traditional motor rehabilitation programs have been beneficial at the onset of this condition to limit the complications of paralysis and with ongoing persistent motor impairments.[28] However, these rehabilitation programs are often tedious and monotonous, leading to decreased adherence. In order to combat the low adherence, a novel virtual motor rehabilitation system has been implemented in two single case studies to assess the effectiveness as an alternative treatment method. An Active Balance Rehabilitation (ABAR) system has been developed for to assist with balance and gait disorders, which utilizes different Virtual Environments to emphasize weight transferences and specific movements that can target symptoms of GBS while addressing other motor impairments. The characteristics of the ABAR system include: A flexible system for the recovery of postural control and for reducing fractures and the risks of falls. A suitable system that improves the patient’s motivation and treatment adherence. A reinforcement system that allows the results obtained in each session to be monitored and the appropriate action taken. A robust system that is able to make a good recovery in parameters such as balance, postural control, muscle tone, and stability in the standing/sitting position in Guillain-Barré patients. A portable system that can be used at home to reinforce the acute and sub-acute stages. A customizable system that offers multiple levels of difficulty that are based on the patients’ progression.[28] Difficulty levels for weight-transferences/movements in the ABAR system.[28] There are two difficulty levels (low and medium) for the ABAR system, with six different games. This allows for various parameters to be adjusted based on patient specific need including level of difficulty, number of virtual sessions, rest period between virtual sessions, session time, target speed, and target display time. The low difficulty level consists of two virtual environments specific for sitting training, allowing for participants to perform medio-lateral and antero-posterior weight transferences in the sitting position. The medium difficulty level consists of four virtual environments that focus on standing training, based on static or dynamic balance rehabilitation. The two patients with GBS participating in this novel research design completed three sessions per week for a total of 20 rehabilitation sessions, consisting of 30 minutes of traditional rehabilitation followed by 30 minutes of virtual motor rehabilitation using the ABAR system. The patient’s demonstrated improvements in static and dynamic clinical balance tests following the ABAR intervention, with more significant improvements demonstrated in static clinical balance tests. In conjunction with the treatment plan outlined above, this virtual motor rehabilitation framework can assist with traditional rehabilitation interventions by enhancing gait and balance outcomes in an engaging manner that will maintain adherence. The difficulty levels and games depicted in the table above focus on simple functional movement tasks to target static and dynamic balance, which will translate to improved movement and gait patterns. This technology is useful in the sense that it can be incorporated into any clinical practice or environment setting, including at home for patients. This system is portable with minimal equipment involved, which can allow for treatment use in areas including private clinics or hospitals. The portability of this treatment can provide allow patients in the acute phase that have been admitted to a hospital to receive effective treatment to limit some of the complications and progression of GBS. This will present a unique in-patient approach that can supplement traditional motor rehabilitation to enhance patient outcomes. In contrast, accessibility to this form of technology may be difficult since it is not as commonly used as a treatment intervention. Although this technology is portable and can be easily integrated into various environments, there can be difficulty acquiring it depending on its availability. The use of this technology is relatively new to GBS treatment, meaning that there are many practitioners that are unfamiliar and do not have adequate training or expertise to implement this intervention effectively. Considering the uncommon prevalence of GBS, there is a lack of exposure and widespread use of this innovative technology that can make it difficult for healthcare providers to implement it into their clinical practice. To mitigate this challenge, additional research supporting the effectiveness of visual motor rehabilitation using the ABAR system should be conducted to showcase its efficacy. This can lead to greater awareness and education of its effectiveness in functional improvements for those living with GBS, and in turn result in greater accessibility for this technology. Robotic Rehabilitation- Cybernic enhanced Ambulation: The use of technology such as overhead harness (used in patients with quadriplegia) to enhance balance in ambulation may benefit David once he is able to attain postural stability in sitting and standing. Interactive biofeedback (iBF) Cybernics treatment therapy using Hybrid Assistive Limb device (HAL) which picks up neural signals in the lower extremity and augments force during walking. This may benefit David significantly while he is initially learning to ambulate. An RCT conducted by Nakajima et al published in 2021, studied the use of Interactive biofeedback (iBF) Cybernics treatment therapy- Hybrid Assistive Device (HAL) in ambulation among patients with debilitating neuromuscular disorders. This study showed significant improvement in quality of gait and strength of the lower extremity compared to conventional use of just hoist after 9 sessions. Outcomes were attributed to patient’s motor learning and functional regeneration with HAL. It has shown to improve motor function in the legs in preliminary studies.[29] Another pre-post study conducted by Brinkemper et al published in 2021 evaluated the use of Hybrid Assistive Device (HAL) in improving the quality of gait in addition to functional parameters among patients with spinal cord injury and other neurogenerative diseases. There was a significant improvement in different phases of the gait cycle. After 12 weeks, sub-groups of patients showed increased step length and swing phase promoting the quality of gait [30] . Once David can walk without the use of a gait, we may progress him to jogging on a treadmill with the support of a overhead harness to prevent falls. Outcome[edit | edit source] Response to Complications and Adverse Events[12] Complication Risk Factors/When to monitor Plan Pressure ulcers Prolonged immobility, lack of sensation Frequent inspection of high risk areas (e.g., malleoli, elbows, etc.). Education on pressure ulcers and the importance of frequent position changes. Therapeutic mobilization as frequently as possible. Compression neuropathy Prolonged immobility, lack of sensation Education on the risk compression neuropathy and the importance of frequent position changes. Therapeutic mobilization as frequently as possible. Contractures, tissue shortening Prolonged muscle weakness and immobility Education on pressure ulcers and the importance of frequent position changes. Therapeutic mobilization as frequently as possible. Referrals A crucial portion of the health care system is to keep patients’ health care teams broad and interprofessional. The aspect of referring a patient to other health care professionals allows patients to get a well-rounded treatment and care plan, centered around their needs. It has been demonstrated through an overview of studies following GBS patients for 6 months, 1 year and 2 years following their diagnosis, that symptoms can widely range from severity of disability following discharge. [31] Respiratory Therapist For David, a respiratory therapist would be the first and prioritized referral. About 2 weeks following David’s admission into hospital, he started showing respiratory distress, which culminated in him being intubated. Respiratory failure begins to develop during the progressive stage in about a quarter of GBS patients, usually resulting in transfer to the ICU and being mechanically ventilated[1]. A respiratory therapist would be extremely beneficial for David as they can provide professional advice and treatment to help with current ventilation ability as well as any further prevention of ventilatory complications. Monitoring David’s use of accessory muscles, breathing depth, inspiratory and expiratory muscle fatigue/atrophy, coughing ability/strength. Drawing blood for monitoring his oxygen levels. Education on proper breathing strategies for both the David and his support system Proper breathing techniques after extubating. Making sure David knows to move arms (within his ability) throughout the day to avoid contractures. Providing reassurance to David that around 80% of patients who are mechanically ventilated make extensive recoveries within their first year after onset.[12] Occupational Therapist A second referral for David would be to an Occupational Therapist. David is currently unable to ambulate due to bilateral lower extremity impairment as well as sensory and motor loss to upper extremity as well. An occupational therapist’s role is to assist a patient in the activities of daily living (ADLs), help their home environment become more accessible, fitting and prescribing certain aids that may help people with daily activities (gait aids, reaching objects, sticky mats, etc.). OT would assist David with functional exercises that will help him independently carry out ADLs and IADLs. With the addition of resistance training if able[32] Assist David with fine motor control of the hand, work will be affected as he uses a mouse throughout the day. Help David with sensation in upper extremities, in turn will also contribute to fine motor skills as well. Provide education on possible reorganization of household items or rooms to accommodate gait aid as well as making ADLs easier to carry out. Social Worker or Psychologist The third referral being made for David would be to a social worker or a psychologist. David has only been diagnosed with GBS for less than a month, this represents a severe change to his life, lifestyle, and hobbies. David was previously active and loved to spend time with his wife and two daughters, and held a successful job in the software development industry. A psychologist would be very beneficial for David to help him with reassurance, grounding, and provision of mental support around these lifestyle changes. Help connect David with support groups with other patients with GBS and their journeys and experiences. Giving David someone to talk to about this traumatic change in his life. Help David cope with any stress or anxiety initiated by not being able to care for his wife and daughters like he used to. Also, guidance for both David and his wife following this diagnosis, it has been shown that some anxiousness and social dysfunction can occur within family members/relatives[31] Questions[edit | edit source] 1. Select two correct answers below regarding Guillain-Barré Syndrome: Unilateral numbness, tingling, and/or pain begins in the hands and feet GBS is a rare autoimmune disorder that affects the Schwann cells GBS is more common in women than men The incidence of subtypes is similar between countries The most common form of GBS is Acute Inflammatory Demyelinating Polyradiculoneuropathy (AIDP) 2. Select the correct answer: GBS patients should always exercise to muscular failure to reap the greatest benefits GBS patients should have decreased rest breaks through their home exercise program PNF stretching has been shown to be effective for improving motor function and control in patients with GBS GBS patients should avoid isometric exercises due to over-reliance on muscular contractions and not compensatory movements 3. Select the correct answer: Hemiparesis is a common presentation of GBS GBS only affects skeletal muscle GBS patients suffer from Uhthoff’s Phenomenon GBS can be triggered by bacterial or viral infection Acute Motor Axonal Neuropathy - PhysiopediaIntroduction Acute motor axonal neuropathy (AMAN) is classified as a subtype of Guillain–Barré syndrome (GBS)[1]. The disease was first discovered in China and was nicknamed 'chinese paralytic disease'[1]. Pathology in AMAN patients are usually limited to the motor nerve, with macrophages destroying axons while myelin remains intact, a contrast to AIDP GBS (most common form of GBS) which is known as a demyelinating disease[1]. While AMAN mainly involves axonal damage to motor nerve fibres; acute inflammatory demyelinating polyradiculoneuropathy (AIDP) involves both motor and sensory nerve fibre damage due to demyelination[1]. AMAN has an estimated prevalence of 30–65% in Asia, Central and South America[2]. Aetiology[edit | edit source] AMAN is strongly linked to Campylobacter jejuni and Zika virus infection.[3][4] AMAN has some documented association with hepatitis E infection.[5] Pathogenesis[edit | edit source] GBS is a post-infectious disorder with two-thirds of patients presenting symptoms of a respiratory or gastrointestinal tract infection before the onset of GBS[6]. Serum antibodies against GM1a, GM1b, GD1a and GalNAc-GD1a gangliosides are noticeable in patients with AMAN[6]. Studies have also shown complement activation causing nerve degeneration at the nodes of Ranvier and at the motor nerve terminal in a mouse model of AMAN.[7] Clinical features[edit | edit source] AMAN presents similarly in terms of clinical features as well as CSF findings to GBS:[1] Weakness of limbs (earliest symptom is weakness of lower limbs) Weakness of respiratory muscles Gradual weakening and finally, absence of tendon reflexes Bulbar palsy in 61% of patients[1] Autonomic dysfunction and pain[8] However, it generally presents without any sensory symptoms, unlike AIDP-GBS[5], although it can present with sensory symptoms[9]. A neurophysiological screen in patients with AMAN should also show no evidence of demyelination, unlike GBS[5]. Differential Diagnosis[edit | edit source] The differential diagnosis of GBS- AMAN should rule out: infectious diseases malignancy disorders of the neuromuscular junction spinal root inflammation due to cytomegalovirus or HIV transverse myelitis Lyme disease A lumbar puncture is recommended to rule out diagnoses other than GBS- elevated protein level and normal cell counts in the CSF (termed albuminocytological dissociation) is considered a hallmark of GBS. EMG-NCV studies also help to diagnose GBS as well as assist in identifying the subtype of the condition (AMAN, AIDP). AMAN patients tend to show decreased motor and/or sensory amplitudes , and complex neurophysiological findings with transient conduction block or slowing, which rapidly recovers during the course of the disease—a phenomenon called reversible conduction failure.[6] The prevalence of pain can help differ between the subtypes of GBS[10] - many patients with AMAN also experience back and neck pain[11]. Prognosis[edit | edit source] The prognosis of AMAN is worse compared to the other subtypes of GBS[12][13]. Medical Management[edit | edit source] Medical management for AMAN combines multidisciplinary supportive medical care and immunotherapy. IVIg and plasma exchange have pleiotropic immunomodulatory effects and are considered primary treatment measures[14]. Studies have shown that patients with AMAN had better outcomes after plasma exchange than after IVIg therapy, and that plasma exchange was also the most cost-effective option[15]. Economically challenged countries have the option of small-volume plasma exchange or exchange transfusion as a low-cost therapeutic option.[15] Physiotherapy Management[edit | edit source] Physical therapy has shown a lot of improvement with pain management and neuromuscular efficiency. Physical therapy modalities of massage and relaxation, proprioceptive neuromuscular facilitation have shown to have positive results. [16] Additional Details can be found on the GBS page.Case Study: Guillain-Barre Syndrome (Sub-Acute) - PhysiopediaAbstract This case study documents a fictional narrative highlighting the approach to care of a patient presenting with Guillain-Barre Syndrome (GBS). The patient's experience in acute care is discussed briefly and his sub-acute rehabilitation is explored more thoroughly. Overall, this case highlights both the nuances of GBS which lead to individualized treatment as well as management of this pathology is related to similar cases. Introduction[edit | edit source] According to the World Health Organization, Guillain-Barre Syndrome (GBS) is a polyradiculoneuropathy which occurs when the body’s immune system attacks part of the peripheral nervous system[1]. This attack may induce sensation impairment, motor control deficit, and severe pain in affected areas leading to disability affecting the upper and lower extremities. GBS is typically preceded by bacterial or viral infection, vaccination, or surgery. Diagnosis is based on symptoms such as bilateral weakness, rapid progression, and hypo/areflexia[2]. Blood tests are not required to diagnose this pathology. Anyone can be affected by GBS - there have been documented cases of patients male and female aged 18 to 80. Since patients with GBS may present similarly to patients with acute exposure to certain toxins, diabetic neuropathy, and Charcot-Matie-Tooth Syndrome, it is important to rule out these differential diagnoses by monitoring symptom progression and taking a history[3]. Early treatment involves acute medical care because this syndrome could progress to life-threatening lethality. In ICU and acute care settings, physiotherapists have several roles. One is to prevent contractures and other negative side effects of demobilization through positioning, splinting, and active assisted range of motion exercises or passive range of motion exercises depending on patient characteristics[4]. Physiotherapists may also be consulted to administer cardiopulmonary intervention and assessment depending on progression of the disease[3]. As symptom progression plateaus and patients transfer from acute care to rehab, treatment focus shifts to favor training focusing on performing functional tasks. Case Background[edit | edit source] Tom Brown, 56 year old male, first presented to the ER with a severe gastrointestinal infection approximately 5 months ago. After this infection was dealt with medically, he was discharged home with no change in baseline or complications. A few weeks later he felt numbness and tingling in his feet and hands along with pain that got worse with movement. During the following two weeks he proceeded to feel weaker and the tingling progressively worsened, eventually leading to admission to the same hospital’s emergency department. Tom could hardly move for 2 weeks and required ICU medical support. The healthcare team did an excellent job of ensuring he at least attempted to mobilize routinely and was positioned properly to avoid contractures/other complications (using postural drainage techniques, splinting, Active Assisted Range of Motion (AAROM), etc., all performed as necessary). It has been 4 months since his last admission to the hospital. No other medical conditions/comorbidities have since arisen and his medical status has remained stable. Tom’s rehabilitation will employ a graded approach centered around the capacity to perform functional activities. The providers of his treatment will be sure to ensure too much is not done too soon - over-exertion could lead to a flare-up and subsequent regression. Effort will be made to involve Tom and his family members in rehabilitation to enforce patient-centered care at every step. Client Characteristics[edit | edit source] Patient Profile: 56 years old , Male Medical Diagnosis: Guillain-Barre Syndrome Primary Complaint: Pain in feet, weakness in upper and lower extremity, fatigue, lack of coordination Past Medical History: First diagnosed with GBS approximately 4 months ago Hypertension Medications: Amlodipine - 6mg 1x per day Smoker - 5 packs / year for 5 years. Has not smoked in 3 years Drinks casually, 1-2 drinks per night Primary Reason For Referral: Complications with GBS including, characterized by ICF Framework: Inability to perform some ADL’s without support (activity level) Mobility, endurance and coordination levels have not returned to pre GBS levels (activity level) Decline in participation in previously enjoyed social events (participation level) Examination Findings[edit | edit source] Initial Assessment[edit | edit source] Subjective[edit | edit source] Present Condition: At the time of assessment, Tom reports that he has regained some motor control of his upper and lower extremities but he still feels very weak and uncoordinated. Weakness and impaired coordination leads to fumbling while completing daily chores. He also has lost some mobility, in particular with raising his arms and flexing his hip. This has led to difficulty dressing and getting into the shower on his own. He has gained enough motor control back to eat independently; however, this is still a challenging task. He is able to walk short distances, about 10 minutes at a time, with a four-wheeled walker, but must stop due to shortness of breath (SOB), fatigue, and pain in his feet. His sensitivity to pain, in general, has increased notably. He has noticed that once non-painful things such as friction on his skin or muscle contraction now elicit a painful response in, he experiences particularly high levels of pain in his back and thighs. Tom reports feelings of embarrassment from all the help he needs from his wife. He has been seeing a speech-language pathologist manage difficulty swallowing as well as an OT for help making his home more accessible. Tom lives in a 2 story home in the suburbs. His bedroom is on the second floor, which he can access independently thanks to sturdy railings recommended by the OT. However, this is difficult and scary for Tom, so he avoids using the stairs as much as possible. Social History: Tom lives with his wife and teenage son, they have both been supportive in his recovery. Before Tom had GBS, he worked on the assembly line at a car manufacturing plant. His wife is a police officer. Tom’s work has been understanding, however, he has not been able to return to work yet and worries that the pain in his feet will keep him from ever returning to his regular duties. On weekends Tom would get together with his friends at the local pub to play billiards. Since the GBS he has not returned to the pub and misses the enjoyment this brought him. Goals and Expectations: Tom’s primary goal is to be able to get dressed and get into the shower on his own (BSF + activity). He would really like to depend on his family less for such basic tasks. Second to that Tom would like to be able to return to work. Although he is close to retiring, he would like to keep working to support his son through college. Lastly, Tom would love to return to playing pool with his friends. He is worried that if he is not able to play pool he will lose touch with his friends. Tom is hoping physiotherapy can help him increase his upper extremity (UE) coordination, range of motion and strength. He was also hoping to receive some exercises to improve his walking so he can be less dependent on his walker. Lastly, Tom has heard that physiotherapy may also help with his pain but is a little skeptical that physiotherapy has much to offer him in terms of pain reduction. Yellow Flags: Worries about ever being able to return to work. Embarrassment from depending on family. Worried he’ll lose touch with his friends. Does not believe physiotherapy can decrease his pain. Score of 11 on the PHQ-9. Pain: Tom describes his pain as a dull ache after activity and sharp and shooting pain during activity. He finds the pain intensity fluctuates but some pain is always present. The pain is diffuse in nature but is most intense in the feet, followed by the back and thighs. He describes the pain in his feet as a 4/10 at rest and 8/10 when weight-bearing, the pain in his lower back at 2/10 at rest and 6/10 when weight-bearing. and his thigh pain at 2/10 at rest and 7/10 when weight-bearing. The pain is at its worst after a bout of walking and at its best when he first wakes up. However, at the end of the day, he has some difficulty falling asleep due to pain brought upon by friction from the sheets on his skin [4]. Objective[5][edit | edit source] Nero scan: Upon examination, all of Tom’s reflexes were diminished but still present at a low level (1+), besides his knee jerk ( L3 reflex) which was absent (0). No differences between individual dermatomes were observed but general hypersensitivity was found. Additionally, a 10mg monofilament elicited strong pain response before bending on feet bilaterally. A slight drooping of the face is also visible indicating facial nerve palsy (CN V trigeminal - sensory + CN VII facial - motor) [6]. ROM: Tom presents with bilateral rigidity and spasticity in his upper and lower extremities. All Passive movements are limited by mild cog-wheel type rigidity and muscle spasm. Movements at almost all joints are limited to some degree, however, the below movements are very limited and impose the most restriction to Tom’s function. Shoulder Abduction: 69/175 degrees PROM Hip Flexion: 56/ 110 degrees PROM Ankle Dorsiflexion: 5/ 20 degrees PROM Information on ROM: Click here Manual Muscle Testing (MMT): Tom was able to perform all motions tested against gravity, however, he has general weakness present in all muscle groups. The largest functional limitations are due to the following muscles: Ankle plantar flexors (PF) (2+) Hip extensors (2+) Knee flexors (2+) Shoulder abduction (2+) Information on MMT: Click here Balance Assessment: Tom is able to stand on two feet independently with eyes open and completed a Berg Balance Scale (BERG) assessment to measure balance capabilities (see below for details). Gait assessment: Tom was able to ambulate 200m with a four-wheeled walker during a 6-Minute walk test. He walked at a slow pace and had to stop twice during the test. He walks with a narrow and short stride. He has a slight drop foot and relies predominantly on hip flexion and abduction to progress feet forward due to weak plantar flexors. Tom completed a Timed Up and Go (TUG) assessment to assess falls risk and gait. He was unable to perform the task without a 4-wheeled walker [7]. Pulmonary function tests: Tom’s cardiorespiratory function was assessed because GBS is known to cause respiratory failure in 20-30% of cases (Dynamed, 2018). Tom’s maximum inspiratory pressure (MIP) was 80 cmH20 and his maximal expiratory pressure (MEP) was 176 cmH20. A spirometry test was performed to further assess Tom’s cardiorespiratory function and safety to perform exercise (located below). Tom presented with a normal functional vital capacity (FVC) and slightly reduced forced expiratory volume in one second (FEV1), which was a sign of a restriction in expiration capability and respiratory muscle weakness [7]. Table 1: Spirometry for Initial Assessment Measurement Predicted Measured % Predicted FVC (L) 4.08 3.97 97% FEV1 (L) 3.06 2.09 68% FEF25-75 (L/s) 3.23 2.12 65% Clinical Impression[edit | edit source] Problem List[edit | edit source] Pain in feet Weak Ankle plantar flexors Hip extensors Quadriceps and shoulder abductors Limited Range of motions (ROM) in the shoulder abduction Hip flexion and ankle dorsiflexion Inability to dress independently Poor balance and unsteady gait Fatigue Physiotherapy Diagnosis[edit | edit source] Tom is in the sub-acute phase of recovery from Guillain-Barre Syndrome. As a result, he has difficulty with balance and UE coordination. Weakness in the plantar flexors and pain in his feet are preventing him from ambulating further than 200m. UE coordination and limitations in shoulder ROM keep him from playing pool with his friends and getting dressed independently. Intervention[edit | edit source] Tom is a 56-year-old male presenting four months post-hospital admission of onset of Guillain-Barre Syndrome. He has now been referred to the outpatient rehabilitation department of the hospital and is looking forward to starting rehab. Tom is presenting with decreased upper and lower extremity strength and coordination, decreased ability to independently complete ADL’s, reduced balance, and decreased ambulation ability due to deconditioning. He also shows a slouched posture, upper and lower extremity rigidity, pain in his feet, and reduced ROM. He reports he’s goals are to be able to get dressed on his own, play pool with his friends and get around without his walker. The Approach to Tom's care will be to improve upon his presenting features, aim to reach his goals and do so in a way that is important and meaningful to him. Rehabilitation will address a multitude of factors for Tom and attempt to return his function. “As individuals begin to improve, they are usually transferred from the acute care hospital to a rehabilitation setting. Here, they can regain strength, receive physical rehabilitation and other therapy to resume activities of daily living, and prepare to return to their pre-illness life.[6] Tom has recovered from the early stages of GBS and is now ready to begin rehabilitation, “For optimal recovery, a two-phase rehabilitation process should take place—the first in the early stages of recovery to diminish the disability burden and the second in the later stages of the disease to support reconditioning.”[8] The physiotherapy approach to GBS will consist of a graded approach to new stimuli such as gait training, range of motion exercises, endurance, strength and functional activities to slowly improve outcome measures and minimize the chance of fatigue. Treatment will include “maintenance of the patient’s posture and alignment, maintaining joint range of motion (passive, active, active assisted), providing ankle-foot orthosis to prevent plantar contractures, improving endurance (repetitive exercises with low resistance), strengthening different muscle groups, and improving flexibility with a progressive ambulation program."[9] A major part of treatment in GBS is the return to functional activities. Not only can functional training help Tom reach his goals, but it can also help improve motivation, program adherence, and transferability. Functional training can include “safe transfer skills, equilibrium and balance in all positions and progressive ambulation."[10] To help reach Tom’s specific goals, Gait training, balance training, functional ADL training, and a gradual introduction to the pool table will be included in his program. Program Notes[edit | edit source] After assessment of Tom’s baseline ability, we will begin at a clinically reasonable level of intensity, duration, and frequency and progress as warranted. Tom’s rehabilitation will take place in the clinic and can be practiced at home, given proper assistance and technique. All parties involved, including Tom, should be aware of the symptoms of fatigue to prevent rehabilitation from regressing and to promote long-term functional independence. [8] Programs for individuals recovering from GBS tend to last around 12 weeks and include 30–60 minutes of exercise intervention three times per week.[8] Aerobic Training[edit | edit source] Low-level aerobic exercise provided safe and positive clinical benefits for this GBSP.[8] Beginning at a low intensity and build towards moderate-intensity on the Borg Scale of Perceived Exertion Scale[11] Frequent rest periods initially A gradual increase in exercise time Exercise modality could include arm ergometer, stationary bike, or walking and should be adjusted to patient ability and preference. Strengthening[edit | edit source] Include both isometric and isotonic resistance training. Prioritize multi-joint exercises. In the early stages of recovery, “By linking multiple joints together for movement, the patient reinforces muscle synergies where primer movers and stabilizers are active and coordinated.”[11] Examples include and are not limited to clamshells, glute bridges, hip kickbacks, side-lying hip abduction, banded dorsiflexion, sit to stand and resistance band upper extremity training. Balance & Gait[edit | edit source] Parallel bar gait & balance training Tandem stance balance & gait Progression to balance on unstable surfaces ( foam surface etc.) Provide ankle-foot orthosis – if needed/tolerated Assess gait aid ability and progress as needed. Range of Motion & Posture[edit | edit source] Joint range of motion (passive, active, active assisted)[9] Progression to active stretching & range of motion Postural cueing, education, and postural exercises. Functional Training[edit | edit source] Functional training can help connect new strength, range of motion, balance and work towards increasing coordination and skill. Progressive introduction to activities (walking without a walker) ADL training (dressing practice, strategy, and education) Return to pool playing – practice with a pool cue, standing endurance, table leaning, and building towards coordinated pool shoot. Pain Management[edit | edit source] Pain neurophysiology education Transcutaneous electrical nerve stimulation Massage with passive ROM Reassurance and explanation of what to expect can help in the alleviation of anxiety that could compound the pain [12] Reassessment 12 weeks Post-Treatment[edit | edit source] Present Condition: 87% of patients with GBS will make full recovery or have minor deficits within 1-3 years [13]. Tom had been diagnosed with GBS approximately 6 months ago and his symptoms from his original diagnosis have begun to stabilize during therapy, which assisted in his progression. Tom’s rehabilitation program consisted of 30-60 minute treatment sessions for 3 days/week. Simple muscular Strength, ROM, and balance assessments were recorded every 2 weeks to ensure progression occurring. A complete reassessment was completed after 12 weeks of treatment. Facial Palsy and Dysphagia: Tom demonstrated increased facial muscle strength and was capable of controlling his facial movements with ease. Collaboration with a speech-language pathologist and OT contributed to his decreased dysphagia and improved ability to swallow. Subjective - 12 weeks:[edit | edit source] Pain: Tom reports decreased intensity and frequency of the pain in his feet, back, and thighs. However, Tom still experiences dull aches in those areas after completing physical activity and weight-bearing for extended periods of time. His sensitivity to pain from random stimuli decreased slightly, but there were times where it seemed to flair up (objective values located in the table below). Goals and Expectations: Because of his increased functional capabilities, Tom feels less embarrassed compared to before treatment. Tom has been more independent with his ADLs and does not require any assistance for basically all of his activities. Tom has been able to return to more activities at work and has less pain compared to before. His work has placed him on more active duties, but there are still some restrictions. After a long day of work, he still becomes tired and feels some pain, but not as often as before. Tom has started playing pool again with his friends but has trouble holding the cue at times. Yellow Flags: Tom reports feeling less depressed and is excited to be able to return to participating in the activities he enjoys. His family reports that his general mood at home is better and he has been anxious to help contribute again. Objective - 12 weeks:[edit | edit source] ROM: Tom has increased ROM and strength in his upper and lower extremities. The increase in control and endurance in his muscles has allowed him to walk for longer distances without fatigue and complete more of his ADLs independently (objective values located in the table below). Balance: There are instances where Tom requires to hold onto something to increase his base of support while walking without a mobility aid but has not fallen since starting therapy. Tom does report that he feels more balanced and mobile compared to when he first started therapy and has been more coordinated with his movements. A reassessment of Tom’s balance using the BERG (48/56) has demonstrated that Tom’s balance has increased significantly since his initial assessment. His confidence has increased and has reported a decreased fear of falling. Tom still has difficulty with external perturbations and sometimes uses a cross-over compensatory mechanism. Future assessment will require more advanced balance outcome measures for functional activities (objective values located in the table below). Mobility: Tom uses a 4 wheeled walker for travelling outside of his home, but has been comfortable walking around his home without a mobility aid. Tom was able to ambulate 375m during a 6-minute walk test (6MWT) with a 4-wheeled walker and was able to ambulate 64m without a mobility aid. Tom walks at a slower pace without a mobility aid and has a wider base with short strides. He has a slight drop foot and relies predominantly on hip flexion and abduction to progress feet forward due to weak plantar flexors. Walking with a mobility aid allows Tom to correct his gait and not have to focus on his balance compared to using no mobility aid. However, it is more tiring using the walker compared to walking without a mobility aid, which is an indication for Tom to switch to a quad cane as a mobility aid (objective values located in the table below). Cardiorespiratory Endurance: Tom still experiences Shortness of Breath (SOB) with too much activity, but not as intense or frequently compared to before therapy. Table 2: Objective Outcome Measures Across 12 Weeks Measures Initial Assessment (t) t + 6 weeks t + 12 weeks Pain: Feet Low Back Thigh Rest 2/10, WB 8/10 Rest 2/10, WB 6/10 Rest 2/10, WB 7/10 Rest 1/10, WB 5/10 Rest 1/10, WB 4/10 Rest 1/10, WB 4/10 Rest 1/10, WB 4/10 Rest 0/10, WB 4/10 Rest 1/10, WB 3/10 Patient Health Questionnaire (PHQ-9) 11 10 7 Reflexes All reflexes diminished (1+) Absent L3 (0) All reflexes diminished (1+) All reflexes (2+) L3 reflex diminished (1+) Myotomes Normal Normal Normal Dermatomes Decreased sensation over L3 and L4 Decreased sensation over L3 Normal 10g Monofilament 4/10 pain (visual analogue scale - VAS) 3/10 pain 1/10 pain Shoulder ROM AROM: 69 PROM: 91 AROM: 90 PROM: 115 AROM: 130 PROM: 150 Hip ROM AROM: 56 PROM: 82 AROM: 77 PROM: 100 AROM: 90 PROM: 108 Ankle ROM AROM: 5 PROM: 10 AROM: 7 PROM: 11 AROM: 10 PROM: 12 Manual Muscle Testing (MMT) Ankle PF: 2+ Hip Extension: 2+ Knee Flexion: 2+ Shoulder Abd: 2+ Ankle PF: 3 Hip Extension: 2+ Knee Flexion: 3+ Shoulder Abd: 3 Ankle PF: 3+ Hip Extension: 3 Knee Flexion: 3+ Shoulder Abd: 3+ 6MWT 200m with a 4-wheeled walker *fatigue and SOB* 350m with a 4-wheeled walker 64m without a mobility aid 375m with a 4-wheeled walker 90m with out a mobility aid TUG 37sec with a 4-wheeled walker 25sec with a 4-wheeled walker 12.5sec with a 4 wheeled walker 25sec without a mobility aid BERG 33 40 48 GBS Disability Scale 3 3 2 GBS Disability Scale, adapted from Hughes et al. (1978)[edit | edit source] Score Clinical Performance 0 A healthy state 1 Minor symptoms and capable of running 2 Able to walk 10m or more without assistance but unable to run 3 Able to walk 10m across an open space with help 4 Bed ridden or chair bound 5 Requiring assisted ventilation for at least part of the day 6 Dead Discharge Plan[edit | edit source] After completing 12 weeks of rehabilitation therapy, Tom has been able to achieve his original goals and has become more independent with his activities. He has been able to walk for short distances around his home without a mobility aid, been able to complete his ADL’s without assistance, has decreased restriction at work, and has started to play pool with his friends again. Tom’s symptoms from GBS have significantly decreased since starting physical therapy, which has helped his progress and improvement in function. However, there are concerns with Tom’s safety for balance while walking without a mobility aid and inability to perform certain functional tasks at work. Tom can be discharged based on his progression but would benefit from less frequent treatment with a focus on increasing his function for work, balance with walking w/o a mobility aid, and switching his 4-wheeled walker to a cane. A discussion with Tom will allow for a reassessment of his long-term goals and a new plan of care can be developed. Based on his response, a discharge date can be determined. Discussion[edit | edit source] Acute GBS is characterized by a rapid decline in a patient's control over the majority of major muscle groups, including respiratory muscles [6]. In the subacute phase, patients begin to gradually regain motor control [6], however, residual weakness, pain, respiratory symptoms, and decreased ROM can severely impact their ability to participate in their typical daily activities [6]. These common deficits were all observed in Tom's assessment and quantified using various outcome measures and tests such as the 6-minute walk test and the VAS. Tom's deficits resulted in an inability to participate in his usual social routine and perform basic activities of daily living such as getting dressed. This rapid loss of function can be overwhelming for GBS patients. Accordingly, Tom's mental well being was also impacted by his GBS diagnosis as can be seen from his PHQ-9 score of 11 on the PHQ-9. Thankfully, the majority of GBS patients make a full recovery [13]. Therefore, by reassuring your patient and focusing treatment on functional activity, physical therapy has a high potential to improve your patient's quality of life. A 12-week intervention program was developed for Tom based on his goals and the ICF model. Tom's program included strength, aerobic, balance, mobility, functional training, and an early focus on pain management. Upon starting treatment Tom's GBS symptoms began decreasing and he made significant improvements in his overall function. A reduction in pain at rest, improved ROM with upper and lower extremities, more stability with static and dynamic postures, increased motivation, and ability to walk without a mobility aid are a few of progressions that Tom achieved. Increasing Tom's functional capabilities has allowed him to return to work at a higher capacity, participate in recreational actives, and complete ADL's independently. Tom uses a 4-wheeled walker outside of his home, has some balance troubles while walking without a mobility aid, and experiences mild-moderate pain with activity. He would benefit from continued physical therapy with a progression to more functional exercises. Tom is consistent with his exercise routine at home and would be able to attend treatment at a lower frequency. Tom may also benefit from a quad cane mobility aid to decrease the energy expenditure of using a 4-wheeled walker. Based on Tom's progression, there is a good likelihood his functional capabilities will keep improving and hopefully he will reach the point where he is able to return to his lifestyle prior to developing GBS.Guillain-Barré Case Study: Marie - PhysiopediaAbstract This case study illustrates a fictional narrative of a 53years old female patient who develops Guillain-Barré syndrome (GBS) relating to her diagnosis of COVID-19. The case study documents the patient’s initial admission into acute care until discharge after she achieves her short-

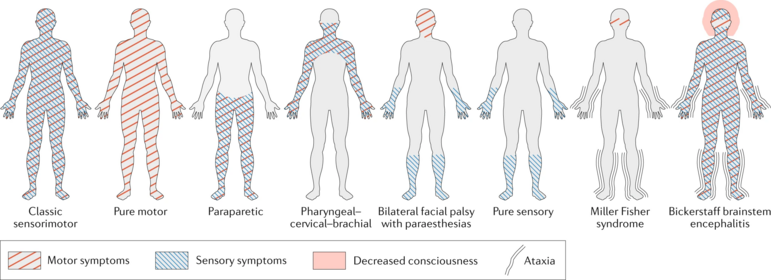
تعليقات
إرسال تعليق